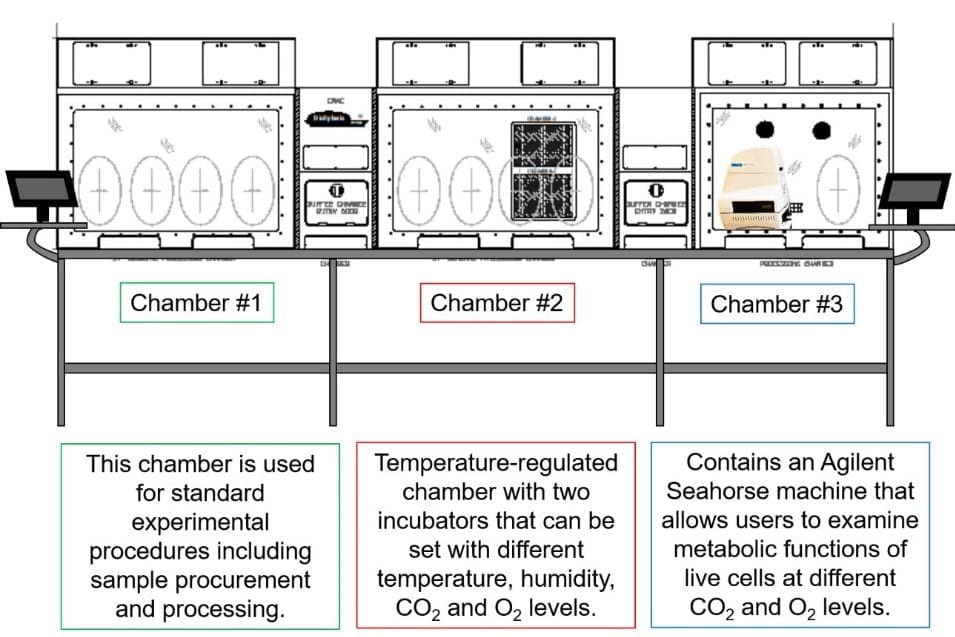
The Hypoxia Core of the IU School of Medicine Cooperative Center of Excellence in Hematology (IU-CCEH) provides resources for members of the IU-CCEH center, other centers, and outside investigators in the area of nonmalignant hematology to generate information critical for better understanding cellular, molecular, and biochemical aspects of hematopoietic stem, progenitor, immune and stromal cells under relevant physiological conditions (i.e., lowered oxygen tension).
O2 tension within bone marrow (BM;1-5%) and cord blood (CB) or mobilized peripheral blood (mPB; <10%), is lower than in the ambient air (~21% O2) in which they are routinely collected for analysis. While it has been known for >40 years that HSC and HPC grow better ex-vivo in lowered (≤ 5%) O2, the core's report (Mantel, et. al., Cell, 2015) and more recent work demonstrate that collection/processing of BM, CB, and mPB in ambient air results in large decreases in phenotypically defined and functional HSC and increased HPC numbers within minutes of cell exposure to ambient air. The core termed this process Extra Physiological Shock/Stress (EPHOSS). Collection/processing of cells at 3% O2, such that they are never exposed to ambient air, resulted in two to five-fold increases in phenotypically- and functionally-detectable HSC. As well, this manifested as different gene expression patterns and responsiveness to stimuli associated with HSC. Similar changes were also noted with murine pre-leukemic and immune cells. Reevaluation of hematopoietic function associated with maintenance of HSC and HPC at lowered O2 levels can now be elucidated through the expertise of the Hypoxia Core. New data derived with the core's hypoxia chamber demonstrates the incredible power of the core to serve the hematopoietic community.
Specific aims of the Hypoxia Core involving normal and nonmalignant murine and human hematology are:
- Provide outstanding, consistent and timely analysis of mouse and human BM, mPB, immune and stromal cells, and human CB, BM, and mPB collected/processed at 3%, as well as other, O2 tensions compared to that in ambient air.
- Coordinate with the other IU-CCEH cores for in-depth analysis of cells collected in hypoxia vs. ambient air.
- Enhance productivity of IU-CCEH and other CCEH members, and outside investigators by providing a centralized set of services with reduced cost, and expert guidance not currently available elsewhere.
- Provide advice regarding intricacies of such studies, analyses, and overcoming potential problems. The core provides an economic resource that will add new and significant cutting-edge scientific analysis to greatly advance our current understanding of HSC/HPC and immune and stromal cell biology in ways that will increase their translational use for clinical benefit.
Services
- Expert guidance on designing experimental protocols.
- Options to have the core staff train individuals to use the equipment or have our trained staff perform the experiment.
- Access to an Agilent Seahorse machine under varying O2 and CO2 concentrations to examine cell metabolism under physiological conditions.