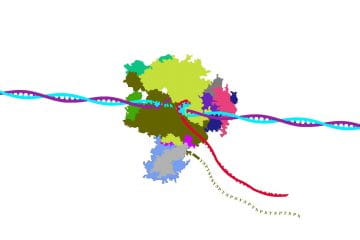
The research lab of Amber L. Mosley, PhD, is working on uncovering novel mechanisms involved in the control of RNA Polymerase II transcription elongation and termination. This work is foundational to understanding how transcription is controlled in normal cells and how these pathways are defective in a wide-array of human diseases including cancers, diabetes, and neuropathologies.
Active Research
Overall, the Mosley Lab is focused on projects to characterize the role of the RNAPII CTD in the regulation of transcription elongation, RNA processing, and transcription termination. This team of investigators is uniquely positioned to tackle unanswered questions about the CTD through analysis of the RNAPII interactome and post-translational modification state of the native RNAPII CTD and its associated proteins using a combination of quantitative proteomics, genomics, genetics and biochemical methods.