The laboratory of Xue Yuan, PhD, explores epithelial stem cells. These remarkable regenerative cells maintain and renew tissues throughout the body. We aim to uncover how epithelial stem cells are regulated under normal conditions and during wound healing. Additionally, we aim to understand the alterations that occur in epithelial stem cells and their niche in the context of various diseases, ranging from inflammatory conditions to cancer.
Active Research
Epithelial stem cells and wound healing
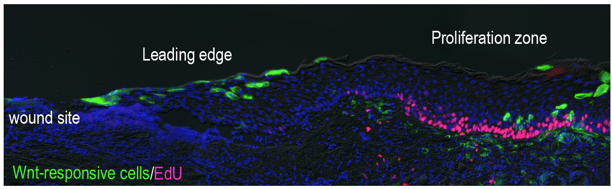
At the core of our research lies the exploration of epithelial stem cells in the process of wound healing. Our earlier investigations revealed the central role of Wnt-responsive cells as epithelial stem cells, essential for maintaining tissue homeostasis and orchestrating their regeneration. While these stem cells are crucial for repair, their regulation remains poorly understood. Fundamental questions persist: What signals trigger the activation of these cells and direct their proliferation and recruitment to injured sites? How is the leading edge formed, and what mechanisms shape the proliferation zone during wound healing? Is there evidence of de-differentiation in suprabasal cells in response to injury, and if so, what initiates this process?
Furthermore, our research extends its focus to the behavior of epithelial stem cells and their niche in the context of diabetes-associated tissue inflammation. Diabetes, often associated with chronic inflammation, presents a unique challenge for regenerative processes. Our studies delve into the adaptation and behavior of epithelial stem cells in these complex environments, seeking to uncover the mechanisms that enable them to function optimally even in the presence of ongoing inflammation.
By shedding light on the regulation of epithelial stem cells during wound healing, both in healthy tissues and in chronically inflamed states, we can gain a better understanding of these essential drivers of regeneration. This knowledge can ultimately be harnessed to improve tissue repair outcomes in a variety of disease contexts.
Post-healing tissue remodeling and stem cell attrition

The majority of stem cell research has centered around their role in the wound healing process. However, there has been limited exploration of stem cell dynamics after the tissue has fully healed. Our research has unveiled a significant insight: even though wounds may heal rapidly, the remodeling of post-healing tissue continues for an extended period. Remarkably, we've observed distinctive patterns in the distribution of Wnt-responsive cells within the healed epithelial tissue. Our primary focus lies in understanding the regeneration of the epithelial stem cell niche within this healed tissue and comprehending the distribution of these cells. Additionally, we're intrigued by the origin of these newly emerging stem cells and how they retain inflammatory memory, potentially playing a role in the long-term maintenance of tissue health. These concepts open up exciting avenues for further investigation.
Head and neck squamous cell carcinomas (HNSCCs) and cancer stem cells
 As our lab delved deeper into investigating the stem cell transformation to malignancy, we made the groundbreaking discovery that Wnt-responsive cells possessed the potential to become cancer stem cells, initiating the formation and metastasis of HNSCC. In response to oral mucosal injury, we observed a surge in Wnt-responsive cell populations, followed by a return to normal levels after the re-epithelialization process. This self-pruning seemed to act as an intrinsic tumor suppressive mechanism.
As our lab delved deeper into investigating the stem cell transformation to malignancy, we made the groundbreaking discovery that Wnt-responsive cells possessed the potential to become cancer stem cells, initiating the formation and metastasis of HNSCC. In response to oral mucosal injury, we observed a surge in Wnt-responsive cell populations, followed by a return to normal levels after the re-epithelialization process. This self-pruning seemed to act as an intrinsic tumor suppressive mechanism.
 Our current research focuses on uncovering the protective mechanisms that prevent stem cell transformation. We are investigating the intricate communication between immune cells and epithelial stem cells during wound healing and tumor initiation. Immune surveillance likely plays a key role in eliminating precancerous stem cells. We're also exploring the relationship between wounds and tumor formation, shedding light on an overlooked area - how the body's own healing safeguards can prevent cancer by clearing damaged stem cells after repair. Overall, our work will reveal new facets of the complex interplay between epithelial stem cells and immune cells and unlock new preventive approaches to stop cancer before it takes hold.
Our current research focuses on uncovering the protective mechanisms that prevent stem cell transformation. We are investigating the intricate communication between immune cells and epithelial stem cells during wound healing and tumor initiation. Immune surveillance likely plays a key role in eliminating precancerous stem cells. We're also exploring the relationship between wounds and tumor formation, shedding light on an overlooked area - how the body's own healing safeguards can prevent cancer by clearing damaged stem cells after repair. Overall, our work will reveal new facets of the complex interplay between epithelial stem cells and immune cells and unlock new preventive approaches to stop cancer before it takes hold.

