The research lab of Mark H. Kaplan, PhD, is broadly interested in cytokines and how they regulate inflammatory diseases including allergies, asthma, cancer, and autoimmunity. The labs works at multiple levels, from examining gene regulatory mechanisms and chromatin modifications, to defining how factors control cellular function and inflammatory disease in mouse model system, to finally trying to take observations into human and patient samples to demonstrate the link between basic research and human immune-mediated health and disease. The laboratory’s multi-level approach, performing studies on transcription factor-dependent gene regulation, defining transcription factor-mediated changes in cell function in vitro and in vivo, and ultimately showing conservation in human samples and where possible, linkage to human disease, continues to be a strength in its studies.
Get Research Updates
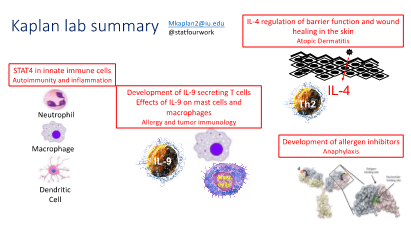
Active Research
STAT Protein Function in T Helper Cells and Allergic Inflammation
A large portion of work in the lab has historically focused on understanding how STAT proteins regulate the development of T helper cell differentiation programs. Over the last 25 years of the program, the lab has defined STAT4 target genes during Th1 differentiation, determined mechanisms of STAT4-dependent gene activation including examining chromatin modifying enzymes and DNA methyltransferases, defined in vivo functions of STAT4-dependent responses in inflammatory disease and pathogen immunity. The lab has characterized STAT4 isoform function in vivo and demonstrated altered expression of STAT4 isoforms in patients with inflammatory bowel disease. The lab has defined how STAT4 integrates with other transcription factors that promote the development of Th1 cells.
Most recently we have developed mice that are conditionally mutant in STAT4 and have ongoing projects examining the role of STAT4 in macrophages and dendritic cells in autoimmune inflammation.
Atopic Dermatitis and skin immunity
Another area of interest is atopic dermatitis, allergic skin inflammation. About 20 years ago a graduate student in the lab developed a mouse model transgenic for a constitutively active STAT6 in T cells. These mice are predisposed towards spontaneous allergic inflammation, particularly in the skin. The lab has characterized the pathogenesis of skin inflammation in this strain focusing in two areas. The first uses this mouse model to test the interactions between the hyperactive Th2 immune response and dysfunction in the barrier function of the skin. The second approach in this area is to define how Th2 cytokines, specifically IL-4, impact keratinocyte gene expression and differentiation. We are using RNA-seq and ChIP-seq approaches to define how the IL-4/STAT6 pathway function in non-immune cells. We have identified that an atopic environment slows skin wound healing and most recently we have identified an IL-4-dependent regulatory circuit that controls the number of gamma-delta T cells in the skin and their ability to participate in the wound healing process.
IL-9 secreting Th9 cells
Among the most recent subsets of T cells described, Th cells that are primed for production of IL-9 are still very poorly understood. We have made significant progress in understanding the development and function of Th9 cells. We demonstrated that PU.1 is a major transcription factor in the development of IL-9-secreting T cells, and that PU.1-expressing T cells are important for the development of allergic lung inflammation in several mouse models. This project has focused on PU.1 and over the past ten years the lab has demonstrated the ability of PU.1 to function as a switch factor, both repressing Th2 development as it increases Th9 development. We published microarray studies of Th9 cells and found several genes that suggest unique functions of Th9 cells. The lab expanded work in this area to examine other PU.1-related ETS factors in the development of Th9 cells, and to examine other transcription factors including BATF and their contribution to IL-9 production and Th9 function. We recently found that STAT5 plays a critical role in opening chromatin to allow BATF to function.
Using a memory model of allergic airway inflammation, we defined a population of IL-9-secreting T cells that remain in the lung tissue long term and provide rapid recall to subsequent allergen challenge. We demonstrated that IL-9 is critical for the recall response and has effects on multiple cell types in the lung. These IL-9-secreting T cells are polyfunctional and secrete other pro-allergic cytokines. Current projects in the lab are focused on defining transcription factors and cytokines that regulate the development of multi-cytokine-secreting resident memory T cells.
We also have several projects related to understanding the function of IL-9, particularly in the lung environment during allergic inflammation and lung cancer. We recently defined mechanisms of IL-9-dependent mast cell accumulation. We also recently defined macrophage populations are important IL-9 responding cells and identified an IL-9/interstitial macrophage/Arginase 1 pathway that is important in the development of allergic lung inflammation and for tumor progression in the lung. Current projects in the lab are focused on more clearly defining the macrophage subsets responsible for IL-9 responses.
Treating immune-mediated disease in an allergic specific approach
The lab is collaborating with a biochemical engineer at University of Notre Dame who has developed novel reagents for blocking immune reactivity in an antigen-specific manner. We have used this approach to block model allergen- and penicillin-specific allergic responses in mouse models. Most recently we have developed peanut-specific inhibitors to prevent fatal allergic response in patients. We have developed a humanized mouse model to assess the ability of the inhibitor to block human mast cell-mediated anaphylaxis. We also hope to use this humanized mouse model to examine human mast cell reactivity to allergens in more detail. The inhibitor approach will be tested with other allergens and autoantigens in the future.
Research Funding
Major Goals: The proposed experiments will provide an assessment of the efficacy of the allergen-specific heterobivalent inhibitor molecules to block anaphylaxis induced in human cells in an in vivo context.
Project Number: 203807IU/ R01 AI108884
Name of PD/PI: Bilgicer
Project/Proposal Start and End Date: 2/10/2020 - 1/31/2025
Major Goals: The goal of the Immunology and Infectious Disease Training Program (IIDTP) at Indiana University School of Medicine is to develop diverse, multi-faceted scientists capable of responding to the nation's health and educational needs in the areas of host immunity and infection.
Project Number: T32 AI060519
Project/Proposal Start and End Date: 8/1/2019 - 7/31/2024
Major Goals: Define the development and function of IL-9-secreting Trm cells.
Project Number: R01 AI057459
Project/Proposal Start and End Date: 11/16/2020 - 10/31/2025
Major Goals: The goal of this project is to define a re- epithelialization regulatory circuit that involves IL-4, γδ T cells, keratinocytes, and fibronectin.
Project Number: R01 AI095282
Project/Proposal Start and End Date: 2/1/2018 - 1/31/2023
Recent Publications
Fu Y, Wang J, Panangipalli G, Ulrich BJ, Koh B, Xu C, Kharwadkar R, Chu X, Wang Y, Gao H, Wu W, Sun J, Tepper RS, Zhou B, Janga SC, Yang K, Kaplan MH. (2020) STAT5 promotes accessibility and is required for BATF-mediated plasticity at the Il9 locus. Nat. Comm. 11(1):4882. PMC7523001
Mehrpouya-Bahrami P, Moriarty A, de Melo P, Keeter WC, Alakhras N, Nelson AS, Hoover M, Barrios M, Nadler JL, Serezani H, Kaplan MH, Galkina EV. (2021) STAT4 is expressed in neutrophils and promotes antimicrobial immunity. JCI Insight. 6(14):141326. PMCID: PMC8410094 (Co-corresponding/senior author)
Fu, Y, Pajulas A, Wang J, Zhou B, Cannon A, Cheung CCL, Zhang J, Zhou H, Fisher AJ, Omstead DT, Khan S, Renauld JC, Paczesny S, Flavell RA, Takatsuka S, Kitamura D, Sun J, Bilgicer B, Sears CR, Yang K, Kaplan MH. (2022) Pulmonary interstitial macrophages are required to mediate the pro-tumorigenic effects of IL-9. Nat. Commun. 13:3811. PMC9249769
Fu Y, Wang J, Zhou B, Pajulas A, Gao H, Ramdas B, Koh B, Ulrich BJ, Yang S, Kapur R, Renauld J-C, Paczesny S, Liu Y, Tighe RM, Licona-Limon P, Flavell RA, Takatsuka S, Kitamura D, Tepper RS, Sun J, Kaplan MH. (2022) An IL-9-pulmonary macrophage axis defines the allergic lung inflammatory environment. Science Immunol. 7(68):eabi9768. PMC8991419
Ulrich BJ, Kharwadkar R, Chu M, Pajulas, A, Muralidharan C, Koh B, Fu Y, Gao H, Hayes TA, Zhou HM, Goplen NP, Nelson AS, Liu Y, Linneman AK, Turner MJ, Licona-Limón P, Flavell RA, Sun J, Kaplan MH. (2022) Allergic airway recall responses require IL-9 from resident memory CD4+ T cells. Science Immunol. 7(69):eabg9296. PMC9295820.
Faculty Research Team
Additional Research Team Members
Wenwu Zhang
Jilu Zhang
Nada Alakhras
Abigail Pajulas
Anthony Cannon
Cherry Cheung
Michelle Chu
Maya Krishnan
Nahid Akhtar