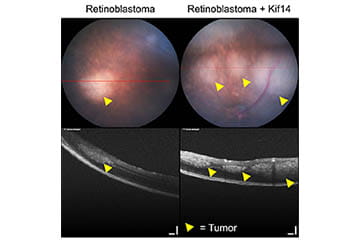
KIF14 is crucial for efficient cytokinesis, although its exact functions remain enigmatic. The Corson Lab research team demonstrated that siRNA-mediated knockdown of KIF14 greatly reduced the proliferation and tumorigenicity of cancer cells in vitro, suggesting its importance for the neoplastic phenotype. Others have since confirmed this in multiple cancer types. The Corson Lab was involved in collaborations to help identify transcription factors and microRNAs that might help drive KIF14 overexpression in ovarian cancer, and to help assess the role of KIF14 in chemosensitivity in triple-negative breast cancer. Recently, the Corson Lab showed that KIF14 overexpression could accelerate retinoblastoma development in vivo, providing key further evidence of this importance of this gene for cancer. The lab continues to explore this and other areas of retinoblastoma biology, including developing novel models of retinoblastoma and methods for analyzing them.
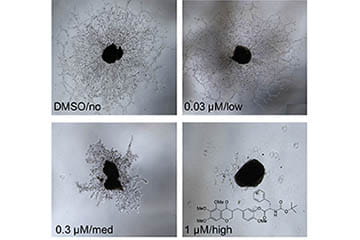 There is a pressing need for novel small molecule drugs to block pathological neovascularization in the eye. In collaboration with Professor Seung-Yong Seo of Gachon University and Professor Dulcie Mulholland of the University of Surrey, the Corson Lab research team are producing novel antiangiogenic compounds based on natural products. The team previously synthesized cremastranone, an antiangiogenic natural product, and developed a novel isomer for this compound with antiangiogenic activity against retinal endothelial cells. More recently, Corson Lab researchers have undertaken a structure-activity relationship study of this class of compounds and developed a new analog, SH-11037, with promising antiangiogenic activity in vitro and in vivo.
There is a pressing need for novel small molecule drugs to block pathological neovascularization in the eye. In collaboration with Professor Seung-Yong Seo of Gachon University and Professor Dulcie Mulholland of the University of Surrey, the Corson Lab research team are producing novel antiangiogenic compounds based on natural products. The team previously synthesized cremastranone, an antiangiogenic natural product, and developed a novel isomer for this compound with antiangiogenic activity against retinal endothelial cells. More recently, Corson Lab researchers have undertaken a structure-activity relationship study of this class of compounds and developed a new analog, SH-11037, with promising antiangiogenic activity in vitro and in vivo.