Bio
Microscopy in 3D is a video about Dr. Bishop's research created by students in the Ball State University Telecommunication Department as a class assignment for Professor James Shasky.
Low Bandwidth Version | High Bandwidth Version
My lab is interested in understanding mechanisms responsible for changes in synaptic connectivity in the nervous system. We study synaptic rearrangements at a model synapse, the neuromuscular junction between motoneurons and muscle fibers, due to its large size, experimental accessibility, and ease of manipulation. Our basic approach is to image synapses from transgenic mice expressing variants of green fluorescent protein (GFP) in their motoneurons using confocal and fluorescent microscopy. We target these same synapses for a much more highly resolved serial electron microscopic reconstruction so that we can directly attribute subcellular alterations revealed by electron microscopy to more macroscopic observations made by light microscopy.
 We are currently using this correlated three-dimensional confocal and electron microscopy for two projects. First, we are investigating a natural process during early development of the nervous system where motoneurons become permanently disconnected from the muscle fibers they initially innervate. At birth, almost every mammalian muscle fiber is innervated by more than one axon. During the next several weeks, all but one axon is lost so that each muscle fiber is innervated by only a single axon into maturity. Since similar processes occur throughout the developing nervous system, we seek to understand the mechanisms involved in sharpening neural connections.
We are currently using this correlated three-dimensional confocal and electron microscopy for two projects. First, we are investigating a natural process during early development of the nervous system where motoneurons become permanently disconnected from the muscle fibers they initially innervate. At birth, almost every mammalian muscle fiber is innervated by more than one axon. During the next several weeks, all but one axon is lost so that each muscle fiber is innervated by only a single axon into maturity. Since similar processes occur throughout the developing nervous system, we seek to understand the mechanisms involved in sharpening neural connections.
A second area of focus involves understanding cellular mechanisms of synapse and motoneuron loss in a mouse model of amyotrophic lateral sclerosis (ALS or Lou Gherig's Disease). ALS is a disease characterized by degeneration of the upper and lower motoneurons that control movement. The protracted loss of motoneurons results in terminal paralysis for which there is currently no cure. 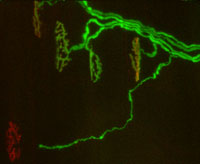
Fortunately, our ability to elucidate the mechanisms of the pathogenesis of ALS has been facilitated by the discovery that a mutant human superoxide dismutase 1 (SOD1) gene found in a subset of human patients with familial amyotrophic lateral sclerosis (fALS) can cause motoneuron degeneration similar to that in humans when expressed in mice. Using several different fluorescent labeling strategies, we have been applying our high resolution imaging techniques to image synapse and motoneuron loss in these transgenic mice in an attempt to understand the subcellular changes that cause or accompany nerve degeneration seen in ALS.
Titles & Appointments
- Associate Dean, IUSM Muncie
- Director, IUSM Muncie
- Associate Professor of Anatomy, Cell Biology & Physiology
-
Education
-
Research
-
Awards

